
Magnetic Beads possess the capacity to fractionate proteins or nucleic acids through the utilization of beads-adsorbent technology that acts as a chromatographic matrix. The process of ion exchange chromatography is prevalently employed for the separation or purification of a target molecule from raw biological materials. The principle behind this procedure is the divergence in surface charges’ accessibility among the molecules, wherein the process utilizes minimal binding and eluting conditions to maintain the biological activity’s integrity.
BcMag™ Weak Cation Exchange (WCX) Magnetic Beads are uniform magnetic beads grafted with a high density of a carboxylate functional group (Fig.1) on the surface. The magnetic bead-based format enables rapid high-yield processing of 96 samples in about 20 minutes. It can quickly fraction proteins or nucleic acids from complex biological samples (such as serum, plasma, etc.) manually or automatically. The purified protein can be used in downstream applications such as sample fractionation for 1D and 2D SDS-PAGE, X-ray crystallization, and NMR spectroscopy. The Weak Cation Exchange resins allow the rapid release of very strong ions that may be retained irreversibly on Strong Ion Cation (SCX) beads. Additionally, weak ion exchangers can be effective separation tools when strong ion exchangers fail because the selectivities of weak and strong ion exchangers frequently differ.
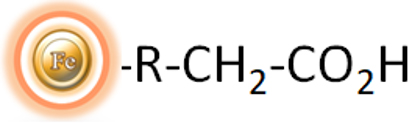
WEAK CATION EXCHANGE (WCX) magnetic beads are used to replace time-consuming, complex, and costly chromatographic procedures such as agarose, cellulose, Sepharose, and Sephadex-based columns or resins. In column-based procedures, the lysate is centrifuged or cleared, the supernatant is added to the column, the membrane or resin is washed with buffer through centrifugation or vacuum manifold, and the required biomolecules are eluted in an adequate volume of buffer. When using column-based technologies, processing multiple samples in academic research labs may necessitate a significant quantity of hand pipetting. This pipetting can discourage differences in target biomolecule yield between experiments and people. Staff and students may require extensive training and practice to produce reasonably constant nucleic acid yields.
Magnetic resins have significant advantages over non-magnetic resin technologies. It is due to the numerous benefits of magnetic beads, such as their ease of use, rapid experimental protocols, suitability, and convenience for high throughput automated and miniaturized processing. They thus see increasing use in various areas of life-sciences research and development, including drug discovery, biomedicine, bioassay development, diagnostics, genomics, and proteomics.
feature and benefits
●
Fast and simple – WCX Magnetic resins-based format eliminates columns or filters or a laborious repeat of pipetting or centrifugation.
●
Convenient and expandable – WCX Magnetic format enables high-throughput processing of multiple samples in parallel with many different automated liquid handling systems.
●
Robust – Magnetic beads do not crack or run dry.
●
Low bed volume – Working with small magnetic bead volumes allows for minimal buffer volumes, resulting in concentrated elution fractions.
Applications
●
Protein pre-fractionation in cell lysates
●
Optimizing purification conditions for new protein preparation protocols
●
Protein purification and concentration
●
Antibody purification from serum, ascites, or tissue culture supernatant
●
Preparation of samples before 1D or 2D PAGE
●
Phosphopeptide purification before MS analysis
Learn More
Instruction Manual
MSDS
Related Ion Exchange Magnetic Beads →General Reference
1.
Wittkopp F, Peeck L, Hafner M, Frech C. Modeling and simulation of protein elution in linear pH and salt gradients on weak, strong and mixed cation exchange resins applying an extended Donnan ion exchange model. J Chromatogr A. 2018 Apr 13;1545:32-47.
2.
Staby A, Jacobsen JH, Hansen RG, Bruus UK, Jensen IH. Comparison of chromatographic ion-exchange resins V. Strong and weak cation-exchange resins. J Chromatogr A. 2006 Jun 23;1118(2):168-79.
3.
Ludewig R, Nietzsche S, Scriba GK. A weak cation-exchange monolith as stationary phase for the separation of peptide diastereomers by CEC. J Sep Sci. 2011 Jan;34(1):64-9.
4.
Chen X, Tolley HD, Lee ML. Weak cation-exchange monolithic column for capillary liquid chromatography of peptides and proteins. J Sep Sci. 2011 Aug;34(16-17):2063-71.
5.
Pabst TM, Antos D, Carta G, Ramasubramanyan N, Hunter AK. Protein separations with induced pH gradients using cation-exchange chromatographic columns containing weak acid groups. J Chromatogr A. 2008 Feb 15;1181(1-2):83-94.
6.
Xu W, Regnier FE. Protein-protein interactions on weak-cation-exchange sorbent surfaces during chromatographic separations. J Chromatogr A. 1998 Dec 18;828(1-2):357-64.



